CAL3QHC and CAL3QHCR are two air dispersion models included in the CALRoads View software package. These models are used to estimate total air pollutant concentrations near highways from both moving and idling vehicles. The original design of the models accounted for carbon monoxide (CO) and particulate matter (PM), but they lacked support for other inert gases. A feature of the brand-new CALRoads View Version 5 allows users to select other pollutants for analysis.
The focus of CAL3QHC and CAL3QHCR was carbon monoxide. The molecular weight of CO (28 grams/mol) was hard coded inside these models. Molecular weight is important to the models because concentrations of inert gases are reported in parts per million (ppm) as opposed to the more recognized convention of amount per some unit volume (e.g., micrograms per cubic meter [µg/m**3]). In order to calculate the correct volume, the model uses molecular weight to convert the calculated amounts with the below equation:
The focus of CAL3QHC and CAL3QHCR was carbon monoxide. The molecular weight of CO (28 grams/mol) was hard coded inside these models. Molecular weight is important to the models because concentrations of inert gases are reported in parts per million (ppm) as opposed to the more recognized convention of amount per some unit volume (e.g., micrograms per cubic meter [µg/m**3]). In order to calculate the correct volume, the model uses molecular weight to convert the calculated amounts with the below equation:
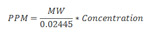
Where: MW = molecular weight [g/mol] and Concentration is given in µg/m**3
Modelers who attempted to use the CO pollutant ID as a surrogate for other inert gases would end up analyzing modeled concentrations that were based on the incorrect molecular weight for their pollutant of interest thus skewing the ppm value.
To resolve this issue, Lakes Environmental recompiled the executables of CAL3QHC and CAL3QHCR to allow for the specification of molecular weight.
Project setup in CALRoads View for inert gases is simple:
Step 1:Open the Job Options window. You can select Data | Job Options from the menu bar or click on the Job/Met icon
Modelers who attempted to use the CO pollutant ID as a surrogate for other inert gases would end up analyzing modeled concentrations that were based on the incorrect molecular weight for their pollutant of interest thus skewing the ppm value.
To resolve this issue, Lakes Environmental recompiled the executables of CAL3QHC and CAL3QHCR to allow for the specification of molecular weight.
Project setup in CALRoads View for inert gases is simple:
Step 1:Open the Job Options window. You can select Data | Job Options from the menu bar or click on the Job/Met icon

Step 2: Select either the CAL3QHC or CAL3QHCR button from the Model selection at the top of the window.
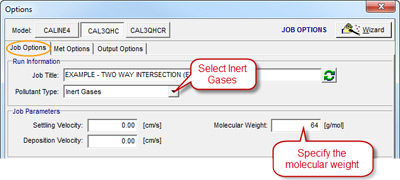
Step 3: The options to change are all located on the Job Options tab of the Options window. First, change the Pollutant Type to “Inert Gases”
Step 4: The Molecular Weight input in the Job Parameters group will switch from a hard coded value to a user input text field. Enter the molecular weight of the gas you are interested in modeling. The example below uses 64 g/mol for sulfur dioxide (SO2).
Step 4: The Molecular Weight input in the Job Parameters group will switch from a hard coded value to a user input text field. Enter the molecular weight of the gas you are interested in modeling. The example below uses 64 g/mol for sulfur dioxide (SO2).
The concentrations produced by the model will now accurately reflect the chemical properties of the pollutant of interest.
Recent Posts


